The Problem

The all-pervasive phenomenon of metal corrosion affects critical infrastructure and vital installations, not to speak of our everyday lives. It leads to
Massive monetary loss estimated at 4% of national GDP,
Structural instability threatening safety,
Indirectly contributes to environmental degradation
Moreover, several studies have indicated mechanical and chemical methods of surface preparation and steel scrappage directly impact carbon footprint. The toxic discharge of corrosion inhibitors and harmful chemical constituents of protective coatings also contribute to environmental disparity.
Electrochemical corrosion happens when metal dissolves (anodic reaction) and corrosion products form (cathodic reaction) in the presence of liquids and air. The environment plays a big role, not just the type of metal. Things like the gases and liquids around, like acids or salt, can affect how corrosion happens. Other factors like humidity, wind speed, and the type of ions in the liquid can make corrosion worse, especially the cathodic reactions.
Problems faced by Existing Coating Solutions
This is not only expensive but will also lead to eventual replacement of painted structures. Zinc based paints/coatings are known to be susceptible to acidic and strongly alkaline attack leading to premature failures in such atmospheres. Even colour-coated galvanized and Galvalume sheets have been found wanting in roofing and cladding applications in industrial installations when exposed to acidic fumes. Extensive surface preparation is absolutely vital for present day protective coatings both on new structural steel as well as pre-painted existing structures. Such preparation ideally calls for grit/shot blasting which is environmentally damaging, tedious, and often unsafe at industrial sites. Similarly galvanized structures (HDG) requireelaborate surface preparation prior to paint application and painted structures are rapidly depleted of zinc in aggressive environments.
Approach to a Solution

Concept of metal passivation
Estimating the extent of corrosion


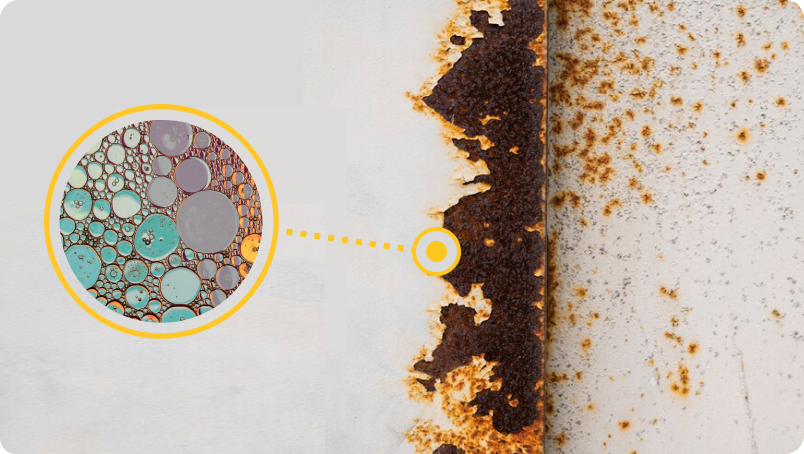
Estimating the extent of corrosion
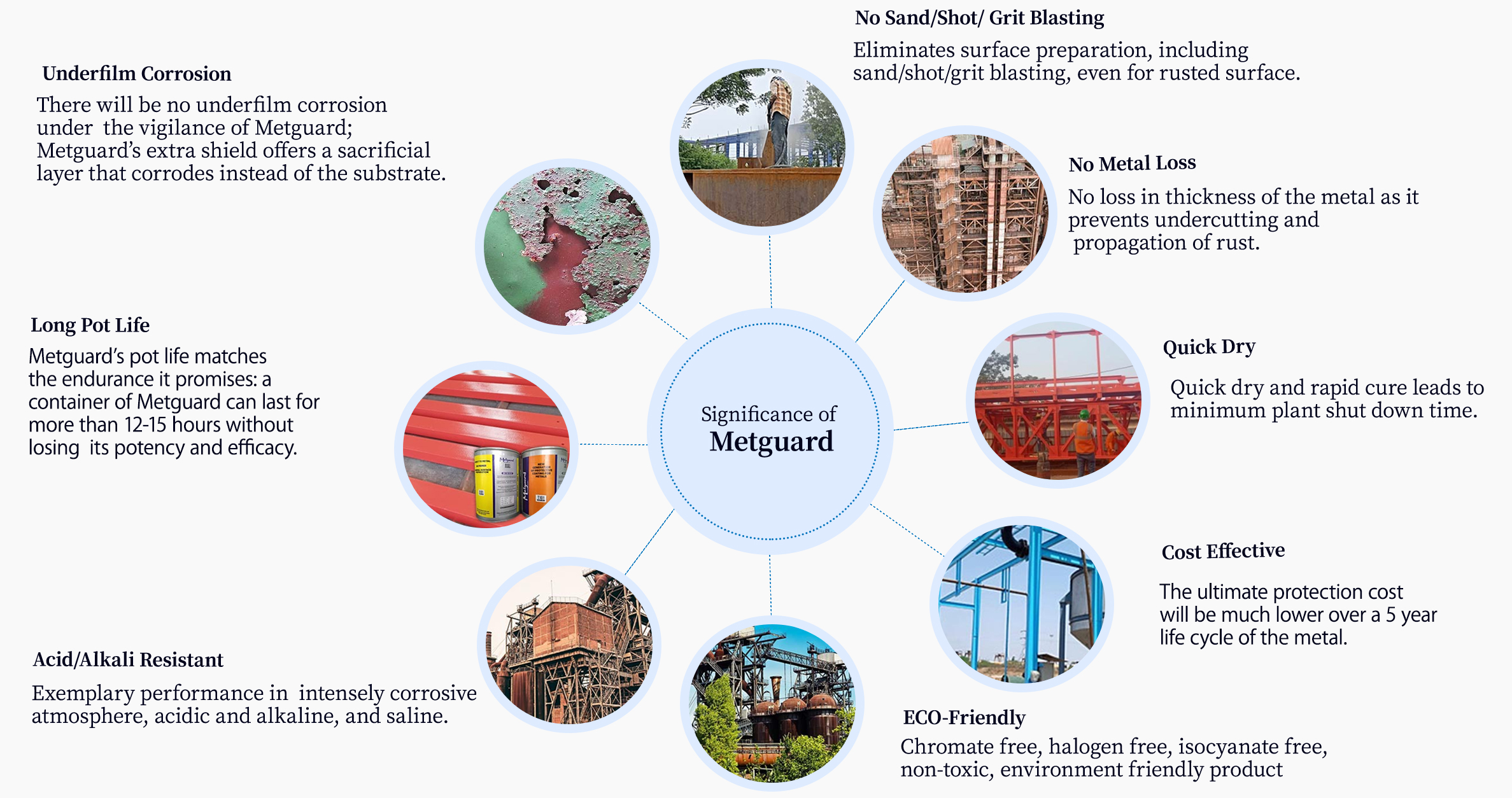
1. No Sand/Shot/Grit Blasting
Eliminates surface preparation, including sand/shot/grit blasting, even for rusted surface.
2. No Metal Loss
No loss in thickness of the metal as it prevents undercutting and propagation of rust.
3. Quick Dry
Quick dry and rapid cure leads to minimum plant shut down time.
4. Cost Effective
The ultimate protection cost will be much lower over a 5 year life cycle of the metal.
5. ECO-Friendly
Chromate free, halogen free, isocyanate free, non-toxic, environment friendly product
6. Acid/Alkali Resistant
Exemplary performance in intensely corrosive atmosphere, acidic and alkaline, and saline.
7. Long Pot Life
Metguard’s pot life matches the endurance it promises: a container of Metguard can last for more than 12-15 hours without losing its potency and efficacy.
8. Underfilm Corrosion
There will be no underfilm corrosion under the vigilance of Metguard; Metguard’s extra shield offers a sacrificial layer that corrodes instead of the substrate.
Advantage Of Metguard

Exceptional adhesion to pre-rusted erected...
Outstanding resistance to dry heat (up to 200°C)...
Toughness and high cohesive strength....
Unmatched resistance to undercutting on steel...
Chromate, isocyanate, and halogen free...
Chromate, isocyanate, and halogen free eco-friendly technology
Application Areas
All industrial infrastructures exposed to hostile emissions located inland or on the coast;
Metalized roofing and cladding panels exposed to acid fumes
Heavy duty transportation, locomotives and rails;
Rebars used in embedded concrete structures;
Application Areas
All industrial infrastructures exposed to hostile emissions located inland or on the coast;
Metalized roofing and cladding panels exposed to acid fumes
Heavy duty transportation, locomotives and rails;
Rebars used in embedded concrete structures;




